By Lawal AbdulMalik-
Scientists have accomplished a World-First, managing to fertilise eggs engineered from the genetic material of adult skin cells in a laboratory setting.
Researchers at Oregon Health & Science University achieved this significant breakthrough, offering immense therapeutic potential for individuals unable to produce viable sperm or eggs.
The new technique, a major step forward in the field of in-vitro gametogenesis (IVG), involves reprogramming ordinary body cells to behave like reproductive cells. This proof-of-concept study demonstrates that creating functional eggs from easily accessible tissue like skin is biologically possible.
This incredible progress, turning human skin cells to eggs, could potentially transform reproductive medicine. The ability to create new eggs from human skin cells to eggs would completely bypass age and illness-related infertility issues. Developing a clinically viable method to convert human skin cells to eggs is the ultimate goal for this research team.
The intricate process began by extracting the nucleus, the cell’s genetic control centre, from a woman’s skin cell. That nucleus contains the complete 46 chromosomes of an ordinary body cell.
Next, researchers implanted this nucleus into a healthy donor egg that already had its own nucleus removed, a technique akin to the somatic cell nuclear transfer used to clone Dolly the sheep.
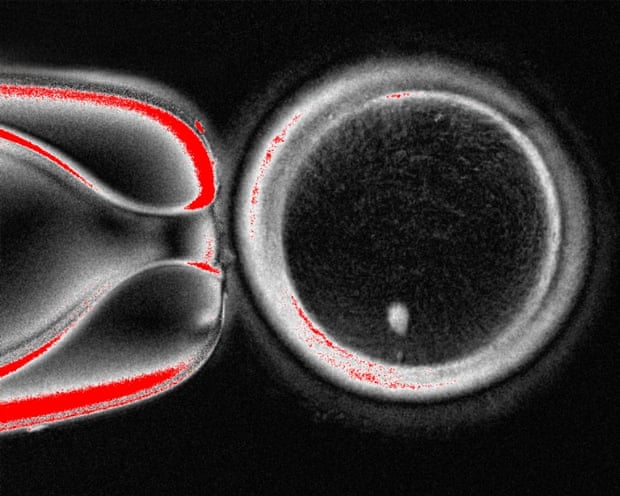
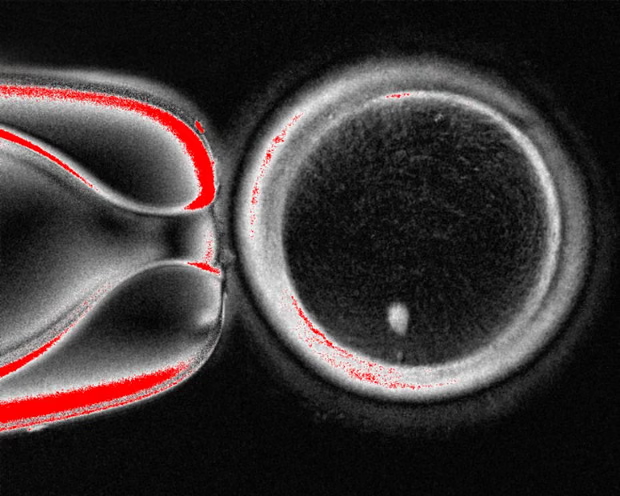
A human egg created through somatic cell nuclear transfer, the process pioneered in the cloning of Dolly the sheep in the 1990s. Photograph: Mitalipov laboratory
The primary challenge involved reducing the chromosome count from 46 to the 23 necessary for a healthy egg.
The team overcame this hurdle by inducing a specialised cell division process they termed “mitomeiosis.” This method mimics natural cell division, successfully causing the cell to discard the extra set of chromosomes. Researchers were able to generate 82 functional eggs, or oocytes, from the process.
Scientific Promise and Remaining Hurdles
Subsequent to their creation, the lab-generated eggs were fertilised with sperm, and approximately nine percent developed into the blastocyst stage after about six days.
The blastocyst stage is the critical point where embryos are typically transferred to the uterus in standard IVF treatment. Despite reaching this important developmental milestone, the resulting embryos showed a high degree of chromosomal abnormalities.
This critical issue means the embryos would not have been suitable for implantation and further development. Scientists acknowledge that while the initial findings represent a remarkable “proof of concept,” a significant amount of research is necessary.
The team estimates at least a decade of further investigation and refinement will be needed to ensure the approach is safe and effective enough for clinical use.
Experts have praised the impressive scientific advance while also highlighting the vital need for caution regarding its application.
Professor Richard Anderson of the University of Edinburgh noted, “The ability to generate new eggs would be a major advance,” but underscored the “very important safety concerns” that must first be addressed
This technology offers hope to millions of people struggling with infertility caused by a lack of viable eggs or sperm.
The technique raises complex ethical questions about the creation of embryos and must be subject to rigorous public discussion and governmental oversight.